Related articles
MedTech Company
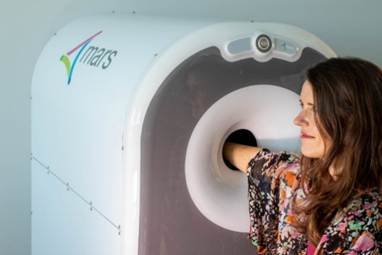
MARS Bioimaging gains FDA clearance for portable photon-counting CT scanner
MARS Bioimaging, a New Zealand–headquartered medical device company, has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its portable photon-counting CT scanner for upper extremity imaging.
.png)
Start-ups
Democratising medicine with a new approach to blood tests
Your GP orders a blood test. Off you go to a lab testing facility. The results come through and your GP asks to see you to follow-up. Do you live near a lab? What if you live deep in the rural sector? Can you get to a lab before or after work? Do you need time off work? When can you fit in another talk with your GP? Can you afford to pay your GP twice?

Orion Health founder Ian McCrae launches AI health-tech start-up, including new tool to stop deadly prescription errors
A start-up says its AI software can save New Zealand’s health system billions of dollars through better patient management – and address deaths caused by prescription errors, which it puts at “road toll” levels.
